What this website is about is the methodology of science, not the opinions of scientists. All real science is a methodology, not an opinion. The most significant methodology of science is mathematics. So the starting point of this criticism is mathematics which proves errors in physics.
The errors began with the misdefinition of kinetic energy in Joule's measurement of 1845. There hasn't been an iota of correct physics produced since then.
Joule stirred water in a wooden bucket for an hour or two (not stated) and supposedly measured the temperature increase. He didn't have the slightest ability to conduct such an experiment, as the slight amount of heat produced would have disappeared into the environment as fast as it was produced. Then there were no correct theoretical concepts for his purpose.
So-called renewables as solar and wind power put more carbon dioxide into the atmosphere than fossil fuel-based energy sources. That's because ungodly amounts of metals are required to produce and transmit the electricity over long distances, while 70-90% of the energy in electricity is lost to heat before it is used for a purpose; and another 60-75% is usually lost in being applied to a purpose.
Where is the science for the claims? It doesn't exist. It's no more than looking out the window. The IPCC makes a stab in the dark and calls it science. Science is a method and analysis with describable knowledge, not the farce created by power mongers imposing fraudulent science and technology onto society.
But isn't the climate changing? It always changes—sometimes heating and sometimes cooling but always moving toward the next ice age which is scheduled to begin at any time. Other Factors Heat The Planet
Physics Summary
Biology Summary
These are the major examples of physics errors described here:
Kinetic energy was incorrectly defined with Joule's measurement in 1845. Joule's Constant
Relativity has no relationship to objective reality. It is used as a control tactic only. Relativity Fraud
Plank's constant was a misinterpretation of electron motion. Quantum Mechanics
There is no significant energy in hydrogen fusion, because Einstein erred in paralleling the erroneous equation for kinetic energy (KE=½mv²). Nuclear Fusion
- The science for greenhouse gases was contrived through fake math. Radiative Transfer Equations
The Stefan-Boltzmann constant shows 40 times too much radiation at normal temperatures. Stefan-Boltzmann
The mechanism for ATP synthesis was scrambled for no reason but promoting corruption. ATP Synthesis
Bureaucrats claim electric motors get 96% efficiency, which is beyond stupidity. Fake Efficiency
About 70-90% of the energy in electricity is lost before being applied to a purpose and another 60-80% is usually lost being applied to a purpose. Electricity Problem
There is not enough metal on planet Earth to convert 10% of society's energy to renewable form and power electric vehicles. Renewable Energy
No one can get past 15% renewables as solar and wind due to fluctuations which are too rapid and long-term to control. Wall At 15%
Electric vehicles get 5 to 10 miles per gallon equivalent, not 100 to 150. Electric Vehicles
Gravity wave measurements were faked—claiming to measure motion at 100 million times smaller than the vibration of atoms. Gravity Wave Measurements
Absurdities Of Greenhouse Gases
One molecule in 2,500 in the atmosphere is never going to have the slightest influence over climate.
Carbon dioxide and other so-called greenhouse gases only absorb long-wave radiation, not the sun's radiation. The long-wave radiation is only emitted by cold substances such as the surface of the Earth. Almost no radiation is emitted from cold substances for "greenhouse gases" to absorb.
Nothing traps heat in the atmosphere. The air cools 20 degrees or more during nights.
The rationalization that nothing but humans could be creating climate extremes is absurd. Other Factors Heat The Planet
An ice age occurs every 100 thousand years, which is an extremely short amount of time. Ocean temperatures drop 5°C; and sea levels drop 400 feet. Then heating occurs for 20 thousand years, while oceans rise.
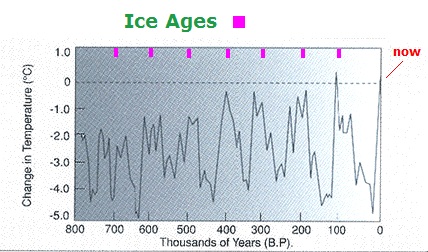
The planet constantly heats between ice ages; and nothing but ice ages cool the planet back down.
Nonscientists assume they can look out the window and determine what science is. They are trying to convince someone that climate is changing. No one needs to be convinced of that. Climate always changes. Then they assume humans have to be the cause. Looking out the window doesn't tell them that. What they don't understand is that any time climate changes, it's not going to be humans that are the cause. Quotes
Thirty years ago, the arguments were somewhat normal in disagreeing over the obscurities of science. But since then, know-nothings in journalism and organizations imposed their claims onto the public and disallowed any criticism. Any scientists who tried to do corrections were silenced or shoved out of science.
How Scientists Are Fired And Intimidated
Physicists calculate radiation using the Stefan-Boltzmann constant. It shows about 40 times too much radiation given off by normal temperature matter. You could heat a house by putting ice on the floor, if the Stefan-Boltzmann constant were correct. Reduce the radiation available to carbon dioxide by a factor of 40 and the whole concept of greenhouse gases would be a joke.
Fraud is overwhelming society, as know-nothings impose their ignorance onto everyone assuming consensus to be fact with no criticism allowed. Consensus is a power mongering tool used by frauds to defy objective reality.
So moral criticism of fraud is a necessary extension of this web site. Without moral truth, the position of corrupters is that there cannot be errors in science, because science is self-correcting.
Fraud in physics set the standard for power mongers taking over the social structures and converting them into power structures for personal gain at everyone else's expense. Trump was the result, not the cause, of the breakdown of social structures and government. The March To Fascism
Renewable energy, electric vehicles and self-driving replace science with magic. It's futurism magic which ignores the failures and absurdities with supposed answers in the future. Without a methodology, the fraud is unlimited.
The IPCC is a fraud organization imposing a greenhouse effect upon society with no methodology that can be verified. Interpretations of weather are translated into a carbon dioxide effect by the frauds.
Incompetents in physics have the same problem as Trump and QAnon: They align assumptions on motives instead of applying rationality to the complexities of objective reality, while the motive of incompetent corrupters is to acquire power through fraud.
Relativity has no relationship to science or the laws of the universe; yet it is imposed upon all physicists with no dissent allowed. In fact, relativity serves as a test of allegiance to fraud, just as street gangsters must commit crime to prove their loyalty.
That's why know-nothings in journalism and organizations can impose fake science and technology onto society with no criticism being allowed. They speak for frauds in physics.
The methodology is the difference between fraud and rationality. Criticism is inseparable from rationality and is always constructive and necessary. Disallowing criticism normalized the fraud standard in society which Trump exploited to replace democracy with totalitarianism. Trump is on track to overwhelm the next election due to increasing fraud and criminality in social standards.
Fraud began in physics with Joule's measurement in 1845. Kinetic energy was misdefined by the result. Ninety percent of physics is corrupted by the error.
Einstein paralleled the error, squaring the velocity of light, which multiplied the energy of fusion almost infinitely. There is no significant energy in fusion.
Quantum mechanics says light is both a packet of energy and a wave—an admitted contradiction but supposedly necessary for the mathematics to work. That's not science. Max Planck's analysis was wrong, because he missed the nature of light acting upon electrons, where a large number of waves hitting an electron looked like a packet of energy.
Out of that deteriorating well of ignorance is where assumptions about greenhouse gases came from. It happened because physics deteriorated into a corrupt power structure which replaced science. Any real scientists who significantly criticize are shoved out of science (Firing Scientists).
Fakery is all physicists produce anymore. Gravity waves are a recent example. Gravity wave measurements supposedly measured motion at 100 million times smaller than the vibration of atoms.
That's like measuring one inch with a ruler that vibrates between Chicago and Los Angeles. The fraud was maximized, so all critics could say is that it is fraud, while the word fraud is not allowed in science or journalism.
There are 2,500 air molecules (nitrogen and oxygen) surrounding each carbon dioxide molecule in the atmosphere. That means the CO2 molecule would have to increase 2,500°C to create an atmospheric temperature increase of 1°C—an impossibility.
These quotes are at the top of almost every state and local web site on climate showing how nonscientific assumptions are imposed upon the public:
...adding large amounts of heat-trapping gases to the atmosphere... There is no such thing as trapping heat in the atmosphere. The atmosphere cools 20°C or more on clear nights due to radiation emitted into space.
...a delicate balance of heat-trapping gases in the atmosphere... Does this graph look like a delicate balance for carbon dioxide?
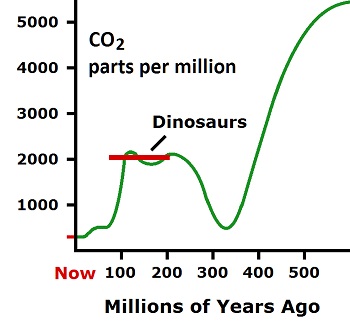
There is not the slightest possibility of one molecule in 2,500 influencing the atmosphere, no matter what its temperature is or what it does. Divide anything carbon dioxide does by 2,500 and it's irrelevant. (Radiative Transfer Equations)
Other factors heat the planet. Climate is always changing; and it is never due to greenhouse gases. Solar intensity has been increasing, as it does every few centuries.
The concept of greenhouse gases was not produced through real science. It originated with incompetents in science, imposed onto the public by nonscientists and then rationalized with fake measurements.
There hasn't been an iota of correct physics produced since Newton's Laws in 1687, because physics is so abstract that it allowed incompetent power mongers to prevail over real scientists. (Origins Of Science Errors)
Physicists began lying 178 years ago to conceal their ignorance. Then they embellished the lying to glamorize their incompetence. Now they contrive fakery to increase their corrupt power, such as a helicopter on Mars, when there is only 1% as much atmosphere on Mars as on Earth, which prevents a helicopter from functioning.
Global Warming
Heat moves into the atmosphere in large amounts through conduction, convection and evaporation, not heating by carbon dioxide. That's why desert air is hot and ocean air is cold, while carbon dioxide is the same everywhere.
Absorbed energy is converted to heat; and heat is radiated away. Radiant energy is weak; but so is absorbed energy. So the energy carbon dioxide absorbs is radiated away as fast as it is absorbed, besides the fact that the quantities are too miniscule to be relevant.
The sun's energy gets into the atmosphere after being absorbed by the surface of the Earth and then being conducted and evaporated into the atmosphere with no more than a slight trace being radiated. No significant amount of the energy is radiated from the Earth's surface into the atmosphere for carbon dioxide to absorb, because cold substances produce almost no radiation.
 Physicists are hung up on an observable absorption dip for carbon dioxide in the Earth's emitted radiation. They missed the fact that the absorbed energy is re-emitted at broadband frequencies which disappear into other frequencies on the graphs. Heat always radiates at broadband, while CO2 absorbs in narrow bands.
Physicists are hung up on an observable absorption dip for carbon dioxide in the Earth's emitted radiation. They missed the fact that the absorbed energy is re-emitted at broadband frequencies which disappear into other frequencies on the graphs. Heat always radiates at broadband, while CO2 absorbs in narrow bands.
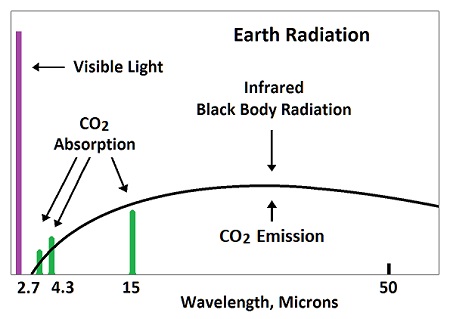
Nothing about a greenhouse effect is based on science, research or measurement. It's based on infantile assumptions by incompetent power mongers who took over physics. Measurements show that saturation precludes the whole concept; but characteristically, the QAnons of physics aren't going to be stopped by measured evidence.
The errors accumulated over time and never got corrected, because science is invisible to the public. But now, the errors are destroying electrical and transportation systems.
What incompetents missed is that it takes more energy to extract energy from the atmosphere than is produced, because wind energy is extremely dilute and concentrating it is energy intensive. The low hanging fruit is already gone, while the alternatives are unreachable and unacceptable. Solar energy is only available 40% of the time in 40% of the locations.
Also missed is that transmission lines hundreds or thousands of miles long are needed with wind and solar energy, while they cost more than the sources when about 300 miles long. There isn't enough metal on planet Earth to get the required energy transmitted through the lines.
The motive for corruption is power mongering, which requires defeat of truth and rationality. Corrupters assume power improves functionality. They are wrong about that. All it does is give corrupters the ability to prevail, while they destroy products and purposes. It puts incompetent corrupters in control of our lives.
| |
Fraud in Science and technology increases day by day in the attempts of incompetent power mongers to prevail over rationality. Democracy cannot be maintained while losing science and technology to fraud.
So-called renewable energy and electric vehicles hit a wall of contradictions years ago but are still portrayed as the wave of the future in exploitation of fraud.
Real scientists and engineers were shoved out of the process, while criticism was blocked and nihilism prevailed. It's always the methodology that produces fraud and its consequences. How Scientists Are Fired And Intimidated | |
|
| |
The Intergovernmental Panel on Climate Change (IPCC) is not science. It is designed by politicians for political purposes with the result being dictated by politicians.
The few scientists involved do no more than contrive scientific legitimacy where there is none. Any real scientists who get involved either quit in disgust or are kicked out for criticizing the process. |
Today's temperature is 5.77λR2/dθ3T4,
becoming λR/755·T/γ3θ by tomorrow.

