 |
|
Research Photo Pages
|
There are two types of evolution. Minor changes occur in a slow and continuous manner with all species. Major changes are made in large leaps which occur rapidly. Both are caused by changes in environmental conditions. Radiation and chemicals produce point mutations for slow change, while flowers and sexuality create large leaps through redistribution of genes. Morel mushrooms are undergoing rapid change, as a yeast growing on trees adapted to the soil. The end point must be a cup fungus for long term survival, because ascospores require that strategy. A cup-like indent is appearing on some variants of the morel, but only a related genus, Helvella, is apt to complete the process. Ascospores are extremely contra-survival for filamentous fungi, which means they only exist when carried from a yeast ancestor. Yeasts and higher fungi evolved in opposite directions creating major differences in many of their important characteristics. Yeasts evolved toward high speed growth through simplicity, while higher fungi sacrificed time for complexity. The basis for the difference is that yeasts must battle more directly against competitors, while higher fungi battle against the elements growing on surfaces in ways which competitors cannot. Yeasts began their evolution about 50 million years ago, as indicated by fossil evidence* and circumstances. Modern plants began producing sugary substances at about that time, which is the primary factor causing yeasts to evolve. Sugar is a dehydrating agent, so it must be in the form of dilute liquid to be utilized by microbes. Since competitors including bacteria and molds can grow in sugary solutions, high speed growth was needed by yeasts. To maximize growth rate, yeasts discarded all possible complexity. Besides simplifying morphology, they minimized the number of enzymes they maintained. Discarded were most extracellular enzymes for breaking down large molecules plus enzymes for using unusual nutrients. Of the remaining enzymes, about ninety percent were repressible being synthesized only when needed. The result for typical yeasts was a mass doubling time of about 90 minutes at room temperature compared to 45 minutes for bacteria. High growth rate was not the only adaptation to increase competitiveness of yeasts in sugary solutions. Also important was the excretion of acid and alcohol. Yeasts normally break down sugars into two-carbon compounds through glycolysis, while repression of TCA enzymes prevents further reduction. Some of the carbon is used for synthesis, but a limited amount is excreted as acid and alcohol to inhibit growth of competitors. The excreted carbon also causes sugar to be depleted more rapidly making it less available to competitors. When the sugar is depleted, TCA enzymes are synthesized, and the excreted carbon is remetabolized. Acid and alcohol excretions by yeasts are under intrinsic control, and the quantities depend upon adaptations. While alcohol must be excreted for anaerobic glycolysis, the extent to which such fermentation is used varies with adaptations. This procedure is effective, because yeasts can develop a high tolerance for acid and alcohol. They usually tolerate pH 3.0 well. Bacteria in general do not tolerate acid as well as yeasts, because their environment is not usually acidic, and because their prokaryotic characteristics do not meet the demands as well as does the complexity of eukaryotic cells. Molds have a low tolerance for acid in organic form, at least when it is absorbed. Circumstantially, the reason appears to be that protons must be pumped out of the cell, while molds must avoid excreting substances which concentrate on exposed mycelium and kill it. The evolution of filamentous fungi traces back in fossil records to the beginning of terrestrial life. New forms of fungi continued to originate from different starting points for several hundred million years. Since yeasts appeared much later, they could only have evolved from higher fungi. The diversity of yeasts indicates that they evolved from several different lines of higher fungi. If this assumption is true, there should be more phylogenetic difference between some yeasts than between some yeasts and some higher fungi. There is a window of opportunity for all evolution; and it closes, because basics cannot change much after they are depended upon. Early in evolution, while basics were still alterable, higher fungi acquired specialized characteristics. Yeasts gave up some of those characteristics and cannot regain them, because that type of evolution no longer occurs. The demanding evolution which higher fungi underwent created the following characteristics:
Yeasts Changed Yeasts not only excrete in a controlled manner but also excrete a variety of metabolites in a manner which appears to be inadvertent. The uncontrolled excretion indicates a lower efficiency of metabolism than higher fungi have. Influencing metabolic efficiency is the spatial arrangement of enzymes which are attached to membranes, and this characteristic would be resistant to change. There is no evidence of filamentous yeasts tolerating dehydration. For example, when insects make holes in trees causing sap to be exuded, the filamentous yeasts grow in the bore holes, where there is protection from dehydration, while unitary yeasts grow in the exudate on the bark. Like yeasts, Morchella mycelium lacks resistance to dehydration. When Morchella is cultured, exposed mycelium dies off and turns brown, unless humidity is saturated. For this reasons, morels only grow in sandy soil or something similar such as mountain humus. Morels follow sandy river basins and become quite isolated due to difficulty getting ascospores out, which results in a lot of localized variations. Sand has the characteristic of not drying out, because it lacks capillary action for moving water to the surface. The morel does not tolerate drying, because yeasts don't. Yeasts evolved from filamentous fungi after modern biology began 65 million years ago. They adapted to sugary solutions produced by modern plants. Fossil evidence shows yeast going back only 50 million years. The significance of these points shows up in the erroneous claim that a leaf mold (Costantinella cristata) is a conidial stage of the morel. Conidia are microscopic stalks with exposed spores on them—the most common way for molds to form spores. Early in the last century, Molliard attempted to grow morels on apple compost in the ground. He inoculated it with morel tissue and covered it with leaf compost. A white sheet of leaf mold covered the surface before a few morels came up. So he studied the leaf mold claiming it was a conidial stage of Morchella. The assumption was totally unwarranted, but it still persists. The leaf mold was said to be found in great abundance on dead leaves in the forest. Morchella could not grow in that manner, because it does not tolerate exposure without dehydrating. Also, it is not a decay organism, as it would have to be to grow competitively on dead leaves. Morphological complexities were also uncharacteristic of Morchella including crosiers and rosettes in addition to conidia. When Morchella mycelium turns brown from dehydration, it excretes alkali, which would primarily be ammonia resulting from residual autolysis. (The alkali is observable on the mycelium with color indicators.) Yeasts and bacteria undergo autolysis upon die off, which allows nutrients to be recycled. Molds do not. Morchella evolved from a yeast so recently that it apparently continues with a degree of autolysis, while it is not advantageous. There is evidence of a disadvantage for Morchella autolysis. Deliquescence is a problem for Morchella. Bacterial attack appears to be involved, as the breakdown is spotty. Also indicating bacterial attack are incidents of long time morel eaters getting sick on morels. The cause in some cases is probably endotoxin from Gram-negative bacteria growing on the surface. Gram-negatives would attack the tissue, while Gram-positives would not; and Gram-positives would be edible. Considering the viscous surface of other mushrooms, they probably promote the growth of Gram-positives by excreting a small amount of carbohydrate. Morchella apparently promotes Gram-negative bacteria by excreting nitrogen through residual autolysis.
There are a number of conclusions that can be drawn from the anomaly with a high degree of certainty. It verifies that Morchella evolved from a yeast, as all other characteristics of the genus indicate. Higher fungi have highly refined morphology which would not allow a rudimentary mushroom to form. Morchella’s lack of strict control over morphology is unprecedented. It could only result from complexities being newly derived and absent in the ancestor. Systematic reproduction of the anomaly under laboratory conditions indicates a pattern which was developed earlier in the evolution of Morchella. The ability to revert to an early evolutionary form indicates very recent evolution. The anomaly is adapted for differentiation on a surface, and therefore it indicates that Morchella started its evolution as a yeast growing on a surface. The surface had to provide long term stability and an interface between above and below ground growth. These requirements would have been met be exudate at the base of trees. It appears that yeasts do not adapt to soil growth. Yeasts are found in the soil, but their prevalence is sporadic indicating that they are there by chance. The main problem for yeasts adapting to the soil is dissemination. Normally, they are disseminated by insects which feed on them and pick them up on their feet. The extent to which air or water disseminates yeast apart from insects is unknown. While the ancestor of Morchella began adapting to soil growth, assuming it was a yeast, it needed to continue with spore dissemination above the ground utilizing insects and eventually wind, before an ascocarp was developed for that purpose. During this phase of its evolution, surface exposure would have created vulnerability to dehydration. Therefore, the weather must have been consistently rainy or very humid. This requirement combined with the timetable of Morchella evolution indicates that Morchella began its evolution at the beginning of an ice age, and ice ages begin with rainy weather. Cold and nonrainy but humid weather might have been adequate, if grass at the bass of trees could prevent dehydration of the evolving yeast. Geologists believe dry and cold weather characterizes ice ages, but perhaps rainier conditions exist at the start of an ice age. Another explanation is that the morel evolved in front of the ice sheet. Cold air sweeping off the ice sheet would have created saturated humidity in a consistent manner. Once a yeast acquired the ability to evolve in the soil, it was drawn in because of the exploitability of bacteria as nutrients. Typical yeasts inherently have the ability to feed on bacteria by killing them with acid, though the quantity of bacteria in their environment is not usually great. The soil contains a large number of bacteria early in the spring due to recycling of nutrients. Freezing provides nutrients for microbes by breaking apart cells and releasing their contents when the ground thaws.
In a study of that species (P.f.), this author found that it undergoes complete die-off sharply below pH 5.0. This pH may be an adaptation designed to release enzymes and recycle nutrients. Regardless of whether it is an adaptation, it means P.f. is highly exploitable by a yeast which can enter the soil and excrete acid. The exploitability of P.f., and perhaps other bacteria, appears to be the primary factor which allowed Morchella to adapt to soil growth in spite of its other characteristics being poorly suited for the purpose. Normally, mushroom mycelium would not be expected to excrete acid, because the acid would concentrate on the mycelium and kill it. Morchella, however, appears to have become dependent upon the excretion of acid for attacking bacteria and feeding on them. To use acid most effectively, it appears that Morchella repeatedly reabsorbs it, channels it through the mycelium and reexcretes it. This mechanism causes the acid to accumulate in absorbent areas while not accumulating on the mycelium. Laboratory evidence of this mechanism is found in nitrogen utilization. In most, if not all, cases, fungi absorb ionic nutrients without their homologous ion, which results in a shift in exogenous pH. When ammonium ions are used as a nitrogen source by Morchella, the medium becomes extremely acidic stopping growth at about pH 3.0. To start growth on ammonium ions, the initial pH must be 6.0 or higher; and even then, mycelial growth is thin. If, however, an alkali creating nutrient is used with ammonium ions, the initial pH can be much lower, and mycelial growth is much thicker. Examples of alkali creating nutrients are K-acetate and Na-glutamate. K-acetate produces a strong rescuing effect for growth on ammonia, because it neutralizes the acid that forms on the surface of the mycelium. Na-glutamate is used more slowly and is thereby less effective. The difference between initial pH and developed pH is that newly inoculated mycelium is not in an ideal condition and must undergo adaptation to the new medium. The adaptations create a heavy drain on the ATP supply. ATP appears to be needed for pumping protons out of the cell but not for absorbing them, as known to be the case with yeast. During initial growth, it appears that protons are freely absorbed but cannot be pumped out because of a shortage of ATP. Therefore, initial growth requires a high pH, or the acidity must be neutralized on the surface of the mycelium through an alkali creating nutrient. Requiring an initial pH of 6.0 for utilization of ammonia indicates a developed tendency for Morchella mycelium to absorb protons as a method of protecting the mycelium from acid. Evidence of Morchella mycelium excreting acid is found in agar media, when using a nitrogen source which does not apparently alter pH, such as casein hydrolysate. The amount of acid produced is proportional to the dextrose concentration indicating that the acid originates with the dextrose, and therefore it would be acetic acid. The amount of acid is not great, as it reduces the pH from 7.3 to 6.0 in typical agar media with moderate buffering capacity. A small amount of acid can be used quite effectively in attacking bacteria by causing it to concentrate where it gets absorbed. So the nutritional evidence combined with the ecology strongly indicate that Morchella attacks bacteria with acid and feeds on them.
Morel ascospores are very large and heavy, containing numerous nuclei each, which causes them to sink in water in a matter of seconds. They therefore travel as projectiles, as demonstrated by the distribution of spores around morels which are drying on a surface. Another limitation to dissemination of Morchella spores is heavy vegetation in the environment resulting in wind currents being diminished and spores being caught by the vegetation. Morels emerge in the spring when there is usually little wind beyond sporadic storms. Also limiting spore dissemination is a failure of morels to come up every year in areas where spring weather can be dry. Considering these factors, spore dissemination is highly unreliable for Morchella, so much so that spores are not depended upon for seasonal survival. Instead, sclerotia serves that purpose. Sclerotia functions as a resistance state for survival of mycelium under harsh conditions. Apparently, modern (gilled) mushrooms must rely upon spores for survival through winter conditions, because the mycelium will not tolerate freezing. Their spores do not usually germinate readily because of their need to survive through the winter. Morchella spores do, however, germinate readily, because they cannot be used reliably for seasonal survival. They are used for dissemination only. Presumably, the ancient mycorrhizals, such as boletes, and puffballs have hardy mycelium which will survive freezing. Truffles might not be so ancient, and the inability of the mycelium to survive freezing may be why they form a mushroom underground.
Emitting spores from the ascus is also problematic for Morchella. Dehydration and shrinkage of tissue create the force which propels spores from the ascus. Often, the tissue does not dehydrate at the right rate. If it dehydrates too fast, the spores will not develop. If it dehydrates too slow, deliquescence will destroy the tissue before spores are emitted. To cope with these requirements, Morchella produces variants which have thick tissue for dry years, thin tissue for wet years and types which are in-between. These variations cannot be genotypic, because natural selection would eliminate all types but one. The differences must therefore be phenotypic only. Apparently, there is no other reported example of such phenotypic variation between individuals which are genotypically the same. However, once the concept is known, it is observable in other species, as described elsewhere on this website. The basic mechanism would be similar to embryonic differentiation, where cells become phenotypically different while remaining genotypically the same. This result is achieved by turning genes on or off in a stable manner. With Morchella, alleles would be turned on or off in a semistable manner allowing remixing of types to occur during each generation. The result is a variety of spore types on a single morel mushroom, which is called "spore strain variation." It has been found that there are identical pairs for each of the different spores, but statistically, there are probably never any spore pairs which are the same. Adapting to environmental needs through phenotypic variation is rudimentary and not highly effective, because less advantageous forms must be produced along with more advantageous ones. However, the mechanism appears to be highly refined, while Morchella is in its evolutionary infancy, which indicates that the mechanism was carried from the ancestor. The mechanism is probably common with yeasts, since they have similar needs for phenotypic variation due to limited gene exchange.
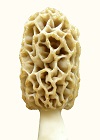
Morels growing in the upper plains of U.S.A. are exposed to hotter and dryer conditions than usual. They have therefore been recently adapting by producing a thicker-tissued variant. Thick-tissued mass has been evolving so rapidly and recently on those morels that it has a tendency to be disordered. This effect shows up as globs of tissue hanging off the cap at the base or oversized ridges creating bumps or globs of tissue. There are thick-tissued morels from other areas which have a more refined morphology.
This indent would be the early evolution of a "cup fungus." It demonstrates the function of the cup which ascosporogenous fleshy fungi usually have. The cup collects water and creates an aggregation of tissue at the bottom while leaving thin and exposed tissue near the rim. The cup thereby maximizes the extremes between slow drying and fast drying tissue. For these reasons, the cup shape is the form toward which fleshy ascomycetes must usually evolve, and there is a convergence of evolution at that point. Of course, the "cup" shape has numerous variations. Morchella would have to evolve into a cup fungus to survive through more than one ice age, which means it is only one ice age old. It appears to restart its evolution at the beginning of each ice age—at least the recent ice ages, which have been cycling at about 100 thousand year intervals for the past million years. The yeast which Morchella evolved from and the ecology would not change much in such a short amount of time; so the same starting point for Morchella evolution appears to redevelop at the beginning of each ice age. There are cup fungi said to be closely related to Morchella based on spore morphology (Nancy Smith Weber, A Morel Hunter’s Companion, 1988). They include Disciotis venosa, Discina leucoxantha and other Discina. Appearing in the spring in an environment similar to that of Morchella but being slightly more advanced than Morchella, they appear to have evolved from the Morchella line during an earlier ice age.
The following are some points of logic indicating that Morchella cannot survive through more than one ice age without evolving into a cup fungus. Morchella is in a state of rapid change resulting in each local environment producing its own variants. Evolution tends to not be repetitive in producing variants; and therefore this state of flux should not have been occurring during the preceding ice age; and Morchella should be in its first ice age cycle. The drastic environmental changes of an ice age should at least eliminate many of the variants of Morchella, particularly since environmental effects have an unusually heavy influence upon its survival, and because its dissemination characteristics are inferior. The Morchella cap is quite impractical for survival under adverse conditions. The cap demonstrates extravagance in producing a large but simple structure which does not tolerate much variation in weather. Such an impractical cap is made possible by the use of sclerotia for seasonal survival, because then ascospores do not have be reliably produced every year. There is much information about the characteristics of Morchella revealed in other mushrooms which evolved with it, which appears to include the families Helvellaceae and Morchellaceae. A very informative example is Helvella crispa. This mushroom is smaller than the morel. It emerges in the same environment as the morel but about a month later, when weather is hotter and dryer. Its cap is shaped like a potato chip hanging over a stalk. Its ascospores do not germinate readily indicating that they are used for seasonal survival, and sclerotia can be assumed to not be produced. Helvella crispa’s small size and absence of sclerotia would stem from a diminished availability of bacteria to feed upon late in the spring. Since it cannot rely upon sclerotia, it must effectively emit spores every year. To achieve this, its cap is much like that of a cup fungus, except that it does not collect water. There is an aggregation of tissue near the stalk, while tissue is thin around the outer edge. Somewhere in-between, drying occurs at the right rate each year. This comparison indicates that Morchella splurges on the shape of its cap, because it can produce sclerotia from the abundance of nutrients which are available early in the spring. But the dependence upon sclerotia limits it habitat to that which can produce a large quantity of bacterial nutrients. The cup fungi have a broader habitat, because they are not dependent upon such rich nutrients for sclerotia production. The cup fungi which are related to Morchella thereby appear to be more similar to Helvella than Morchella, at least in terms of the described characteristics, and therefore they may have evolved from that genus. These types should re-evolve during each ice age along with Morchella. The cup fungi related to Morchella indicate a re-evolution of types with each ice age. During an ice age, a wall of ice a mile thick or more moves south into warmer and dryer climates. In front of the ice is an interface between uninhabitable ice and warm, dry desert conditions. This interface would include all possible environmental conditions packed into a small amount of space. In that small space, humans would find something like a garden of paradise. Having all extremes within easy reach would allow humans to find alternatives for every purpose. This would be ideal for developing agriculture. For some unknown reason, something similar happens for the evolution of the morel mushroom. The morel evolved from a yeast which would have used the base of trees for surface growth while adapting to the soil. There are numerous obstacles to such evolution at the present time. Normally, tree exudate would be needed for nutrients, but it is sporadic. Dehydration would be a problem when there is not a lot of exudate. Perhaps cold, humid air sweeping off the ice sheet creates an improvement upon tree sap at the base of trees. Normally, only elm trees produce a large amount of exudate, and it is sporadic. However, conditions under the loose bark of some trees create suitable conditions for yeasts. The yeast genus Nadsonia has a species, elongata, which is adapted to growing under the loose bark of birch trees. This type of growth may be improved in front of the ice sheet. Note on Ice Ages. The usual description of ice ages is that the whole planet cools for some unexplainable reason. The opposite is true. It's oceans heating that causes an ice age. When warm Pacific Ocean water flows over the Bering Strait, the warm ocean water in the Arctic causes a lot of evaporation of moisture and results in more snow accumulating in the north than can melt during the summers. The accumulated snow reflects away sunlight causing a precipitous cool-down in the north. But since ocean temperatures do not change as easily as atmospheric temperatures, the evaporation continues. The oceans drop 400 ft (130 m) before the reversal occurs. As the oceans drop, increased land mass picks up more energy from the sun. So the land mass gets warm, while the ice accumulates. There is not much north-south mixing of the atmosphere. Therefore, conditions are hot and dry in front of the ice sheet, not cold, as usually claimed. Humans could not have created agriculture during the middle of the last ice age (50 thousand years ago) otherwise. Five indications of Morchella evolving from a yeast are these:
*A reference on fossils:
|

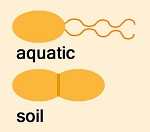 A bacterium which is highly specialized in exploiting cell debris in spring soil is Pseudomonas fluorescens. (We’ll call it P.f. Its name stems from a water-soluble fluorescent pigment which it produces. The purpose of the pigment is to attract insects for dissemination, much like the color of flowers.) It releases proteolytic enzymes into the environment through autolysis, and the enzymes break down proteins from the cell debris.
A bacterium which is highly specialized in exploiting cell debris in spring soil is Pseudomonas fluorescens. (We’ll call it P.f. Its name stems from a water-soluble fluorescent pigment which it produces. The purpose of the pigment is to attract insects for dissemination, much like the color of flowers.) It releases proteolytic enzymes into the environment through autolysis, and the enzymes break down proteins from the cell debris. An unusual characteristic of morels is that they do not measure gravity for growing vertical. Whatever direction they start growing as they emerge from the soil is the direction which they keep growing. Almost everything which emerges from the soil measures gravity for vertical growth. The inability of the morel to measure gravity is additional evidence of its very recent evolution from an organism without macromorphology.
An unusual characteristic of morels is that they do not measure gravity for growing vertical. Whatever direction they start growing as they emerge from the soil is the direction which they keep growing. Almost everything which emerges from the soil measures gravity for vertical growth. The inability of the morel to measure gravity is additional evidence of its very recent evolution from an organism without macromorphology.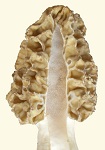
 This means the spores have no ability to be suspended in air without wind. There have been claims of observing clouds of spores over morels, but the basis would be imagination.
This means the spores have no ability to be suspended in air without wind. There have been claims of observing clouds of spores over morels, but the basis would be imagination.


 The morphology of thick-tissued morels is highly informative and must be evaluated in detail. The ascocarp is hollow, which promotes drying. Tissue thickness is controlled primarily by the width of ridges and the amount of space between them. With thick-tissued types, ridges are wide and curved with little space between them. With thin-tissued types, ridges are small leaving much space between them. Another identifying difference is the width of the stem, which correlates with the amount of empty space inside the cap. Thick-tissued morels have a narrow stem, while thin-tissued ones have a wide stem. These factors control how concentrated the tissue mass is and therefore its rate of dehydration.
The morphology of thick-tissued morels is highly informative and must be evaluated in detail. The ascocarp is hollow, which promotes drying. Tissue thickness is controlled primarily by the width of ridges and the amount of space between them. With thick-tissued types, ridges are wide and curved with little space between them. With thin-tissued types, ridges are small leaving much space between them. Another identifying difference is the width of the stem, which correlates with the amount of empty space inside the cap. Thick-tissued morels have a narrow stem, while thin-tissued ones have a wide stem. These factors control how concentrated the tissue mass is and therefore its rate of dehydration.
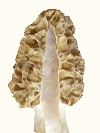
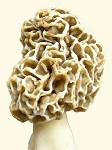 Another feature of the thick-tissued morel is an indent in the cap, which slows the rate of dehydration even further. At the indent, ridges are closer and tissue more concentrated creating a slow-drying area on the cap.
Another feature of the thick-tissued morel is an indent in the cap, which slows the rate of dehydration even further. At the indent, ridges are closer and tissue more concentrated creating a slow-drying area on the cap.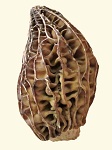 The two morels above are from the upper plains. This morel is from northwestern USA. There is quite often some evidence of the indent in morels, but not always.
The two morels above are from the upper plains. This morel is from northwestern USA. There is quite often some evidence of the indent in morels, but not always.
 Cup fungi vary somewhat in their shape. If they need to dry easily, they have thin tissue; and if they need to delay drying, they have folds in them, as shown in these photos.
Cup fungi vary somewhat in their shape. If they need to dry easily, they have thin tissue; and if they need to delay drying, they have folds in them, as shown in these photos.